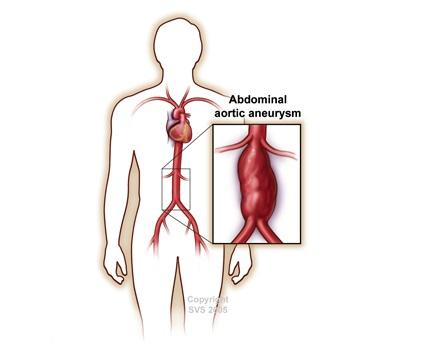
What is an AAA?
The aorta is the largest artery in your body. It starts at your heart and runs through your chest and abdomen. The abdominal area of the aorta is responsible for delivering blood to your legs, gastrointestinal tract, and kidneys. An abdominal aortic aneurysm (AAA) happens when the wall of the aorta weakens over time and begins to bulge like a balloon. If left untreated, an AAA may get bigger and eventually rupture, causing severe internal bleeding and possibly death.
In addition to the risk of rupturing, clots or debris can form within an AAA. These pieces can be carried to other areas in the body and block circulation, which can cause severe pain or even limb loss if blood flow is cut off for too long.
Every year, 200,000 people in the U.S. are diagnosed with an abdominal aortic aneurysm (AAA). A ruptured AAA is the 15th leading cause of death in the country, and the 10th leading cause of death in men older than 55.
Aneurysms often run in families. If a first-degree relative, like a parent or sibling, has had an AAA, you are 12 times more likely to develop an AAA. About 15-25% of people who need treatment to repair an AAA have an immediate family member with the same type of aneurysm.
- No Symptoms: Most people with an AAA have no symptoms and it is often found when tests are performed for unrelated reasons.
- Sudden, Severe Abdominal or Back Pain: If you have a family history of AAA and feel sudden, severe pain in your abdomen or back, seek immediate care. This may signal that you have an AAA and it is possibly rupturing.
- Pain, Discolored Skin, Sores on Feet and Toes: A small percentage of patients have these symptoms when plaque or blood clots from elsewhere in the body collect in the feet and toes.
- Older Age
- Males are more at risk than females
- Family History of AAA
- Smoking
- High blood pressure (Hypertension)
- Atherosclerosis (hardening of the arteries)
- Chronic lung disease
Talk to a Doctor
If you have risk factors for an AAA or have been diagnosed with one, you should talk to your doctor about a referral to see a Vascular Surgeon.
Diagnosis
If an AAA is suspected, your doctor may refer you for:
- An abdominal ultrasound, which is a painless, safe test that screens for and measures the size of the AAA.
- A computed tomographic angiogram (CTA) might also be recommended, which assesses the aneurysm size, location and the extent of impact. It would expose you to radiation and injection of an intravenous contrast agent. However, it provides valuable information that can help your vascular surgeon determine the best type of repair.
Role of a Vascular Surgeon
If you’re diagnosed with an AAA, you should be referred to a vascular surgeon. They can determine when treatment is needed and recommend the best procedure for patients with AAA, based on the size of the aneurysm and other factors.
Because vascular surgeons have specific training in both minimally invasive and traditional open repairs, they can help ensure that the repair is tailored to your unique anatomy and expectations.
Treatment for AAA depends on the size of the aneurysm.
Small AAAs (Less than 5 cm in diameter)
Small AAAs have a very low risk of rupturing and should be watched. It’s important to have an ultrasound test every 6-12 months to check for growth and risk of rupture. Treatment can include:
- Lifestyle changes that help control blood pressure and medication
- Quitting smoking
- Daily exercise
Larger AAAs (More than 5.0-5.5 cm in diameter)
Larger, rapidly growing AAAs and ones causing symptoms are usually repaired. Types of repair include:
Endovascular aneurysm repair (EVAR)
Endovascular aneurysm repair (EVAR) is a less invasive treatment and is most frequently performed.
Your vascular surgeon would make two small punctures in your groin. Guided by X-ray imaging, a tiny device is introduced into the artery. This device is used to reinforce the artery wall and exclude the aneurysm.
Most patients stay in the hospital one to two days. Recovery time is shorter than with open surgery.
Open Surgery
Open surgery is performed to place a prosthetic graft. Your vascular surgeon would access the affected portion of the aortic artery through an incision in your abdomen.
Most patients stay in the hospital four to 10 days. Recovery time may take up to three months.
